INTRODUCTION
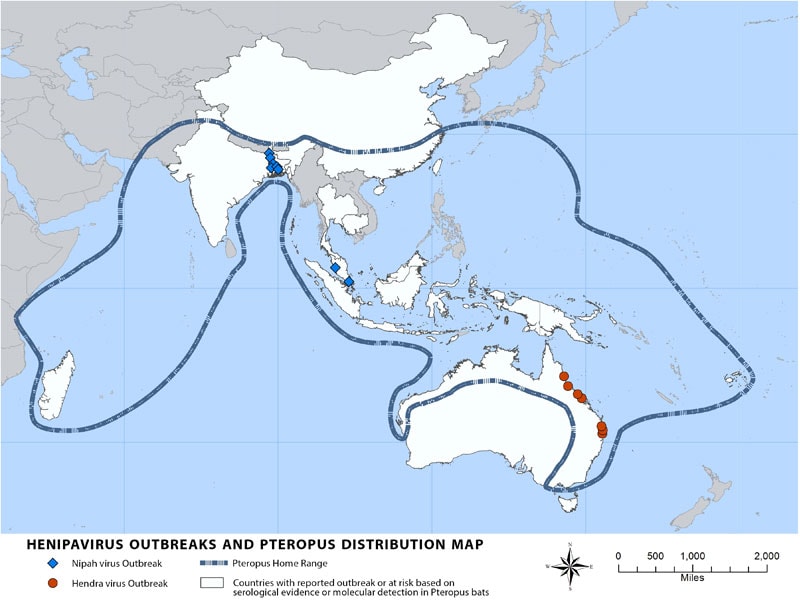
Nipah virus (NiV) is a member of the family Paramyxoviridae, genus Henipavirus. NiV was initially isolated and identified in 1999 during an outbreak of encephalitis and respiratory illness among pig farmers and people with close contact with pigs in Malaysia and Singapore. Its name originated from Sungai Nipah, a village in the Malaysian Peninsula where pig farmers became ill with encephalitis. Given the relatedness of NiV to Hendra virus, bat species were quickly singled out for investigation and flying foxes of the genus Pteropus were subsequently identified as the reservoir for NiV (Distribution Map).
STARTING
In the 1999 outbreak, Nipah virus caused a relatively mild disease in pigs, but nearly 300 human cases with over 100 deaths were reported. In order to stop the outbreak, more than a million pigs were euthanized, causing tremendous trade loss for Malaysia. Since this outbreak, no subsequent cases (in neither swine nor human) have been reported in either Malaysia or Singapore.
In 2001, NiV was again identified as the causative agent in an outbreak of human disease occurring in Bangladesh. Genetic sequencing confirmed this virus as Nipah virus, but a strain different from the one identified in 1999. In the same year, another outbreak was identified retrospectively in Siliguri, India with reports of person-to-person transmission in hospital settings (nosocomial transmission). Unlike the Malaysian NiV outbreak, outbreaks occur almost annually in Bangladesh and have been reported several times in India.
Signs and Symptoms
Infection with Nipah virus is associated with encephalitis (inflammation of the brain). After exposure and an incubation period of 5 to 14 days,illness presents with 3-14 days of fever and headache, followed by drowsiness, disorientation and mental confusion. These signs and symptoms can progress to coma within 24-48 hours. Some patients have a respiratory illness during the early part of their infections, and half of the patients showing severe neurological signs showed also pulmonary signs.
During the Nipah virus disease outbreak in 1998-99, 265 patients were infected with the virus. About 40% of those patients who entered hospitals with serious nervous disease died from the illness.
Long-term sequelae following Nipah virus infection have been noted, including persistent convulsions and personality changes.
Latent infections with subsequent reactivation of Nipah virus and death have also been reported months and even years after exposure.
Treatment
Treatment is limited to supportive care. Because Nipah virus encephalitis can be transmitted person-to-person, standard infection control practices and proper barrier nursing techniques are important in preventing hospital-acquired infections (nosocomial transmission).
The drug ribavirin has been shown to be effective against the viruses in vitro, but human investigations to date have been inconclusive and the clinical usefulness of ribavirin remains uncertain.
Passive immunization using a human monoclonal antibody targeting the Nipah G glycoprotein has been evaluated in the post-exposure therapy in the ferret model and found to be of benefit.
Prevention
Nipah virus infection can be prevented by avoiding exposure to sick pigs and bats in endemic areas and not drinking raw date palm sap.
Additional efforts focused on surveillance and awareness will help prevent future outbreaks. Research is needed to better understand the ecology of bats and Nipah virus, investigating questions such as the seasonality of disease within reproductive cycles of bats. Surveillance tools should include reliable laboratory assays for early detection of disease in communities and livestock, and raising awareness of transmission and symptoms is important in reinforcing standard infection control practices to avoid human-to-human infections in hospital settings (nosocomial infection).
A subunit vaccine, using the Hendra G protein, produces cross-protective antibodies against HENV and NIPV has been recently used in Australia to protect horses against Hendra virus. This vaccine offers great potential for henipavirus protection in humans as well.
